This website uses cookies to ensure you get the best experience on our website.
Read more
FAQs about TEER Measurement
February 02, 2015

Here are some frequently asked questions (FAQ) about TEER measurement using an EVOM2.
- Are the electrical resistance and transepithelial electrical resistance (TEER) the same thing?
- What is an EndOhm chamber?
- How is an EVOM2 used for measuring confluence?
- Why use an EndOhm instead of a STX?
- How do I clean my electrode?
- What about electrode preconditioning?
- Can you give me a simple data acquisition system for TEER?
- What are the TEER measurement challenges that I may encounter?
- Can you suggest some experimental parameters that can be controlled to obtain consistent TEER results?
- I want to measure TEER of alveolar epithelial cells which are cultured without media in the apical side. Do I need to add media/buffer in the apical side to measure the TEER of these epithelial monolayers?
Are the electrical resistance and transepithelial electrical resistance (TEER) the same thing?
No. We obtain the TEER value by multiplying the raw/obtained resistance value on an epithelial voltmeter (such as, EVOM2) by the cell growth area (e.g., area of a cellular monolayer grown onto a cell culture insert).
For example, if you grow cells onto a cell culture insert with area of 0.5 cm2.
The resistance readout on an epithelial voltmeter (e.g., EVOM2) shows a value of 300 Ω.
TEER= 300 Ω × 0.5 cm2 = 150 Ω- cm2
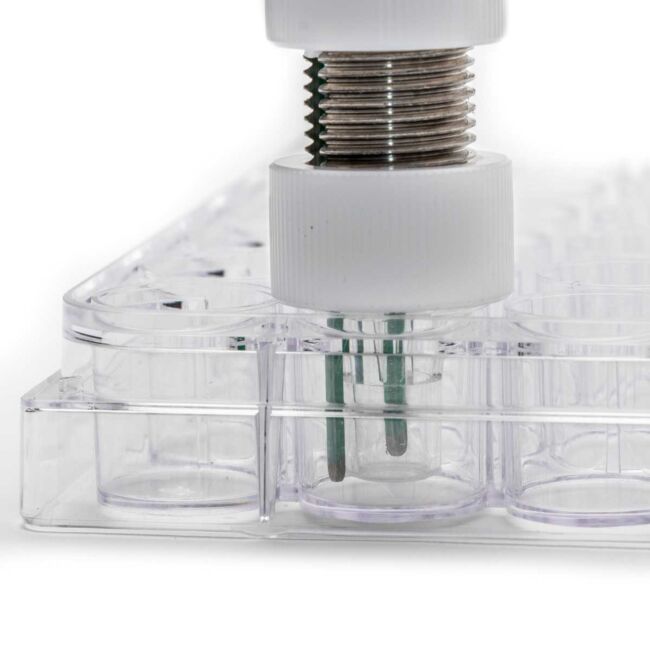
What is an EndOhm?
The EndOhm is a chamber designed for the EVOM2 where you place a removable well into it to make the resistance measurements. This chamber has electrodes in fixed positions and the electrode positioning and stillness has a major affects when measuring tissue resistance.

How is an EVOM2 used for Measuring Confluence?
The EVOM2 works on the principal that once you measure what we call the “blank” well, this first resistance measurement contains the summation of the electrode resistance, the electrode gap and the resistance due to the volume and the molarity of the liquid media. (Any electrode charge differences are negated by the EVOM2's measurement method of reversing the polarity and averaging the results.) The successive periodic measurements of the well are a plot of the growth of the membrane by a resistance measurement, and once this resistance graph has plateaued, we can say that the membrane has reached confluence. The EVOM2 system works just like a Voltage clamp amplifier and Ussing system, but without the special Ussing chamber. The EVOM2 system is not as accurate as an Ussing can be, but the purpose of the EVOM2 system is to to determine if a membrane is confluent, not perform detailed analysis. (Some membrane permeability analysis can be studied by the EVOM2 system, but as a percent of change rather than an absolute value of change.)
Why use an EndOhm instead of a STX?
The EVOM2 will detect pin holes and voids in the membrane above all else as the flow of electricity will seek the lower point of resistance. The STX2 general purpose electrodes can flex and change the spacing of the electrodes, and also the act of not holding them still in a well can add to a significant resistance error. On the other hand, the EndOhm electrodes are fixed in position. When properly cared for and pretreated in media, they give the most stable, repeatable and reliable resistance readings. The addition of the centered concentric electrodes also insure that the entire area of the membrane is exposed to the electrical current flow so there are no shadow areas due to an edge placement as there can be with the STX2. (It should be noted that the baseline expected TEER value of the tissue, if it is of a low resistance value, can add to measurement difficulties with large area membranes. For Example: If the TEER value is 200Ω-cm2 then a 12mm well will be 1.13 times less for the same confluent measurement (176.9Ω). The same 200Ω tissue in a 24mm diameter well is 4.52 times less or 44.2Ω. As this resistance number decreases below about 100Ω, we see more electrode instability and those readings can vary. We have found that electrode “cleaning” and preconditioning are essential for stable readings.)
How do I Clean my Electrode?
Electrode cleaning consists of the periodic use of an enzymatic cleaner (Enzol, Tergazyme) and – or a soak in unscented household bleach (3% NaClO). The bleach serves to recoat Ag surfaces to AgCl and dissolve accumulated proteins. Both of these effects lead to electrode instability. The enzymatic cleaner is a safe agent to remove residue from the electrodes deposited by media components like DMEM. If this is allowed to accumulate, the resistance reading goes up and also can be unstable.
What about Electrode Preconditioning?
Electrode preconditioning is presoaking the electrodes in the measurement media for a time to allow any foreign liquids to migrate out of the permeable pellets. Alcohol is commonly used to sterilize these electrodes and the alcohol will soak into them and act as a high resistance to electrical flow. As the alcohol slowly exchanges with saline based media inside the electrode, the resistance reading results are seen as a downward drifting value.
Can you give me a Simple Data Acquisition System for TEER?
The Lab-Trax-4 is a quiet, 12-bit, four channel, analog-to-digital data recorder that has sampling speeds up to 10,000 samples per second or 2500 s/S for all four channels. This unit is powered directly by the USB2 port and can be used on a laptop in the field as a portable unit. The hardware has four digital inputs and four digital outputs, if needed.
The LabScribe3 software is easy to use and frequently updated to meet the most current demands. As Microsoft continues to update to newer operating systems, the hardware will continue to be supported by newer revsions of LabScribe. It is also now supported on Mac and Linux based computers. If you are using a STX2 to make these measurements, you may need an extra pair of hands to enter text for you. If you enter your text into the Marks field before making measurements, then you only need to press the Enter key to place the mark when your TEER reading has stabilized. If you are using an STX100, you will have your hands free to make notations.
What are the TEER measurement challenges that I may encounter?
This was written to address issues with the STX2, but it also applies to the EndOhm.
Any of these items can change the resistance reading:
- Electrode spacing gap. Do not spread or compress the electrode gap of the STX2 electrode.
- Electrode resistance. Keep the electrodes clean and conditioned.
- Electrode depth. Always submerge the electrodes at least 2 mm.
- Electrodes near the plastic walls. Try to keep the electrodes away from plate walls, if possible.
- Electrode placement. If you typically rest the longer leg of the electrode on the bottom plate, then repeat that placement for consistency.
- Fluid resistance. Do not let the media evaporate and do not dilute it.
- Fluid levels. Try to maintain the same volume of fluid for every reading. A small variation may have a large effect in a small insert.
- Excessive movement. Try to hold the electrodes still. In some cases, a mechanical electrode holder may be used.
- Washing/media change may cause a rupture in the membrane of the cell culture insert. Fluid change should be done cautiously along the wall of the inserts. A small gap of lifted tissue/cellular layer from the membrane of the cell culture insert can open a clear path for the EVOM2 to read a lower resistance. The current leaks through this gap rather than going through the membrane containing the cellular monolayer, since electricity tends to take the path of least resistance. In such cases, you will notice a drastic drop in the resistance. When you notice such a large decrease in the resistance value, we recommend that you check your sample (e.g., cellular monolayer on a cell culture insert) under a microscope to see if there is a rupture in the filter or large empty region on the filter indicating that a cluster of cells has detached.
Acquiring multiple readings in the same well (3-5 times) and calculating the average can be used to decrease the variability.
Can you suggest some experimental parameters that can be controlled to obtain consistent TEER results?
- Temperature is known affect TEER values. We recommend that you maintain a consistent temperature to obtain consistent values. Since the reading are obtained in cell culture media/ buffer. We recommend that you use a water bath with a fixed temperature to warm the media/buffer to be used during the experiment. A consistent media/buffer temperature ensures a consistent experimental condition. We recommend taking the well plate, containing cells grown on culture inserts, out of the incubator for at least 20 minutes to stabilize the well plate at room temperature before making measurements.
- If you are using an EndOhm chamber, make sure you maintain the same fixed distance between the top and bottom electrodes to obtain consistent read outs. If you are using a chop-stick electrode (STX2), try to hold it in a vertical position while obtaining results. Consistency in maintaining the same holding position of the chop-stick electrodes while performing an experiment is expected to show consistent read outs.
- We recommend using the same fluid with the same ionic concentration both in the apical (e.g., top of a cell culture insert) or basolateral side (e.g., lower part of the cell culture insert sitting inside a well of a 12 well-plate). During the measurement, if you are using 1X PBS buffer in the apical side, we recommend using 1X PBS buffer in the basolateral side. We also recommend that both fluid levels (inside and outside of cell culture inserts) be at the same height in order to minimize pressure differentials. During experiments, the apical well/side is filled first with fluid to prevent dislodging of the membrane from the filter by hydrostatic pressures.
- Application of consistent volumes of the fluid (media/buffer) during all experiments will reduce data variability.
I want to measure TEER of alveolar epithelial cells which are cultured without media in the apical side. Do I need to add media/buffer in the apical side to measure the TEER of these monolayers?
Yes. In simple terms, electrodes (e.g., EndOhm/ STX2) maintain electrical connection via fluid (cell culture media/buffer). Not having any fluid on the top of the cell culture insert will disrupt the electrical connection. However, since these cells are cultured without media in the apical side, we recommend exposure to apical fluid for shortest time possible. It is safe to assume that around 5 minutes of exposure to apical media/buffer should not affect the cells. We suggest that you perform a pilot experiment to determine the duration of time that your cells can tolerate apical fluid. As mentioned previously, we recommend using the same fluid with same ionic concentration (for example, 1X PBS buffer or media) in both apical and basolateral sides.
For more information, see Recording TEER Measurements from an EVOM2.

Close