This website uses cookies to ensure you get the best experience on our website.
Read more
Ussing System for Epithelial Research
As low as
$1,350.00
Only %1 left
Prices valid in USA, Canada, and PR only.
Order code
Price range: $1,350 - $2,450

Prices valid in USA, Canada, and PR only.
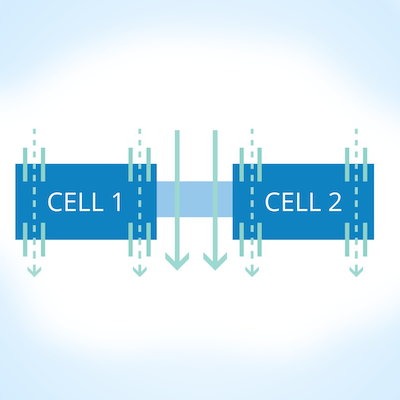
WPI’s classical Ussing Chambers are well established perfusion chambers that are easy to operate, easy to control temperature, and easy to clean after use. Ussing Chambers are machined from solid acrylic with eight entry ports for fluid lines, electrodes, or agar bridges. For easy, leak-free attachment of tubing and electrodes, all eight ports are Luer type. The four ports for voltage and current electrodes are recessed to prevent formation of air bubbles in the chamber. The fluid compartments in each side of the chamber are separated by the epithelial membrane being studied. Sharp stainless steel pins on one side of the chamber hold the membrane in position and mate with holes in the opposite chamber interface. (In the CHM4, tissue is held by an O-ring instead of pins.)
Prices valid in USA, Canada, and PR only.
Non-destructive TEER measurement for epithelial tissue
Features
- Direct connect low-resistance electrodes
- Simple operation, easy to control temperature and clean after use
- Luer type leak-free attachment of tubing and electrodes
- Recessed electrode ports avoid bubble formation
- Secure membrane holding by sharp stainless steel pins or O-ring
- Specialized chamber adapts cell culture insert (Costar Snapwell) for monolayer cell culture
- Chambers with rectangular openings for tubular tissues from small animals
Ussing System Options
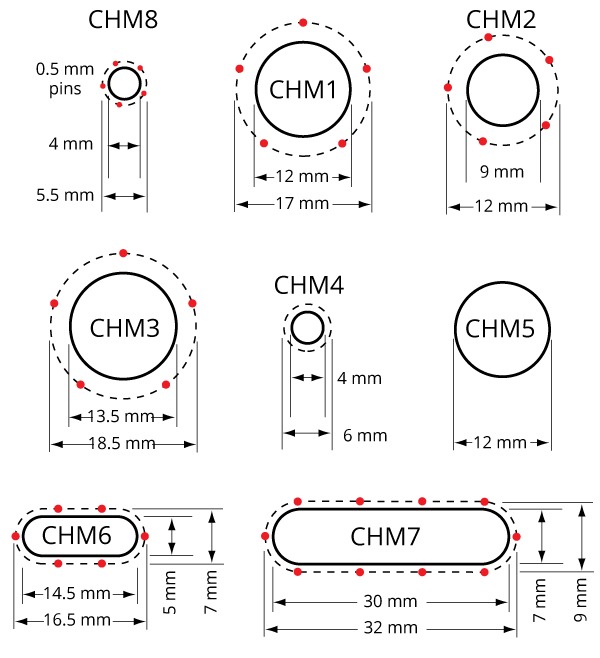
| Order code | Reservoir | Chamber # | Chamber | Reservoir Opening | Half Chamber Volume | Pin Circle Diameter |
| USS1L USS1S |
Large Small |
CHM1 | Medium | 12 mm | 1.0 mL | 17 mm |
| USS2L USS2S |
Large Small |
CHM2 | Small | 9 mm | 0.75 mL | 12 mm |
| USS3L USS3S |
Large Small |
CHM3 | Large | 13.5 mm | 1.2 mL | 18.5 mm |
| USS7L USS7S |
Large Small |
CHM7 | Large rectangular | 7 x 30 mm | 5.5 mL | 9 x 32 mm |
| USS8L USS8S |
Large Small |
CHM8 | Extra small | 4 mm | 0.5 mL | 5.5 mm |
Benefits
- Leak free design of Ussing chambers
- Can be used with monolayer cell culture inserts
- Optional drains for quick evacuation of radioactive or toxic substances
- Circulation reservoirs available in two sizes
- Control temperature with a circulating water bath (option) available
Applications
- Ion transport studies
- Nutrient transport studies
WPI’s Ussing System offers researchers a quick, effective means of making low-resistance electrical connections to the Ussing chamber without need of long agar bridges or Calomel half-cells. Ag/AgCl half-cells screw into short tubes which plug firmly into place in the chamber’s Luer ports. These direct-connect electrodes eliminate the inconvenience and expense of Calomel half-cells in open liquids. The system includes one Ussing Chamber (eight sizes available), support stand, electrode kit, glass circulation reservoir (two sizes available), and a tubing start-up kit (25 feet of 0.375-in. tubing, 10 feet of 0.156-in. tubing, plus four male Luer fittings, two compressor clamps, one Y-connector, and one clip). Sixteen possible system configurations are listed at right. Components are also available separately. (Preamplifier in photo not included.)
Leak free design of Ussing chambers
WPI’s classical Ussing Chambers are well established perfusion chambers that are easy to operate, easy to control temperature, and easy to clean after use. Ussing Chambers are machined from solid acrylic with eight entry ports for fluid lines, electrodes, or agar bridges. For easy, leak-free attachment of tubing and electrodes, all eight ports are Luer type. The four ports for voltage and current electrodes are recessed to prevent formation of air bubbles in the chamber. The fluid compartments in each side of the chamber are separated by the epithelial membrane being studied. Sharp stainless steel pins on one side of the chamber hold the membrane in position and mate with holes in the opposite chamber interface. (In the CHM4, tissue is held by an O-ring instead of pins.)
Can be used with monolayer cell culture inserts
The CHM5 chamber adapts the Costar Snapwell, a cell culture insert for monolayer cell culture, into WPI’s “classical” epithelial voltage clamp system. Classical Ussing Chambers have not been widely used for monolayer cell culture inserts, because most inserts have a very deep profile, limiting good fluid perfusion at the surface of the membrane and limiting voltage electrodes from measuring the potential close to the surface of the membrane. CHM5 solves these problems: Perfusion fluid is introduced into the chamber at an angle so that it flows directly to the surface of the membrane. The voltage electrode is also inserted into the chamber at an angle to reduce the distance between the surface of the membrane and the electrode.
Rectangular openings for tubular tissue
Two small chambers with rectangular openings are designed for tubular tissue from small animals such as the mouse intestinal tract membrane (CHM6) and rat intestinal tract membrane (CHM7). The rectangular opening more closely matches the shape of the tissue than would a circular opening, significantly increasing the membrane area available for testing. The larger membrane area increases the transport rate of low permeability chemicals. It also reduces the electrical resistance of the system for easier current clamping.
Optional drains
Drains may be added to Ussing chambers to allow quick and complete evacuation of radioactive or toxic substances. To have drains added at the time of order, add a “D” to the part number (such as “USS1LD”). The cost of the drain will be added to the cost of the chamber or system ordered.
Cartridge electrodes
The Electrode Kit contains four voltage/current electrodes, plus four Luer-tipped cartridges. Electrodes are threaded and screw securely into the end of each cartridge. The Luer tip then plugs securely into the Luer openings of the chamber. The cable from each electrode terminates with a 2 mm pin which may be plugged into voltage/current clamps such as WPI’s EVC-4000 using the EVC3. The miniature electrode-gel cartridge is a small plastic tube with a male Luer tip identical to those at the tip of hypodermic syringes. The tube may be filled with different gel materials. Agar is commonly used, but other gel materials may also be satisfactory.
Circulation reservoirs available in two sizes
Hand-blown borosilicate glass reservoirs with jacketed chambers for temperature control are available in two sizes:
- #5210 holds 20 mL per side
- #5362 holds 10 mL per side (useful when expensive chemicals are involved).
Reservoir condenser caps prevent air bubbles and turbulence in fluid reservoirs.
Reservoir openings and pins
All systems include the stand, selected reservoir, selected chamber, electrodes and tubing.
| SKU | VAR-3759 |
|---|
McLamb, B. L., Gibson, A. J., Overman, E. L., Stahl, C., & Moeser, A. J. (2013). Early Weaning Stress in Pigs Impairs Innate Mucosal Immune Responses to Enterotoxigenic E. coli Challenge and Exacerbates Intestinal Injury and Clinical Disease. PLoS ONE, 8(4), e59838. http://doi.org/10.1371/journal.pone.0059838
Khera, M., Somogyi, G. T., Kiss, S., Boone, T. B., & Smith, C. P. (2004). Botulinum toxin A inhibits ATP release from bladder urothelium after chronic spinal cord injury. Neurochemistry International, 45(7), 987–93. http://doi.org/10.1016/j.neuint.2004.06.001