Selling fast!
Get yours while you can.
Free Shipping on web orders over $50! (USA only. Some exclusions.) Free Shipping on web orders over $50! (USA only. Some exclusions.)






NANOFIL
Couldn't load pickup availability
Prices valid in USA, Canada, and PR only.
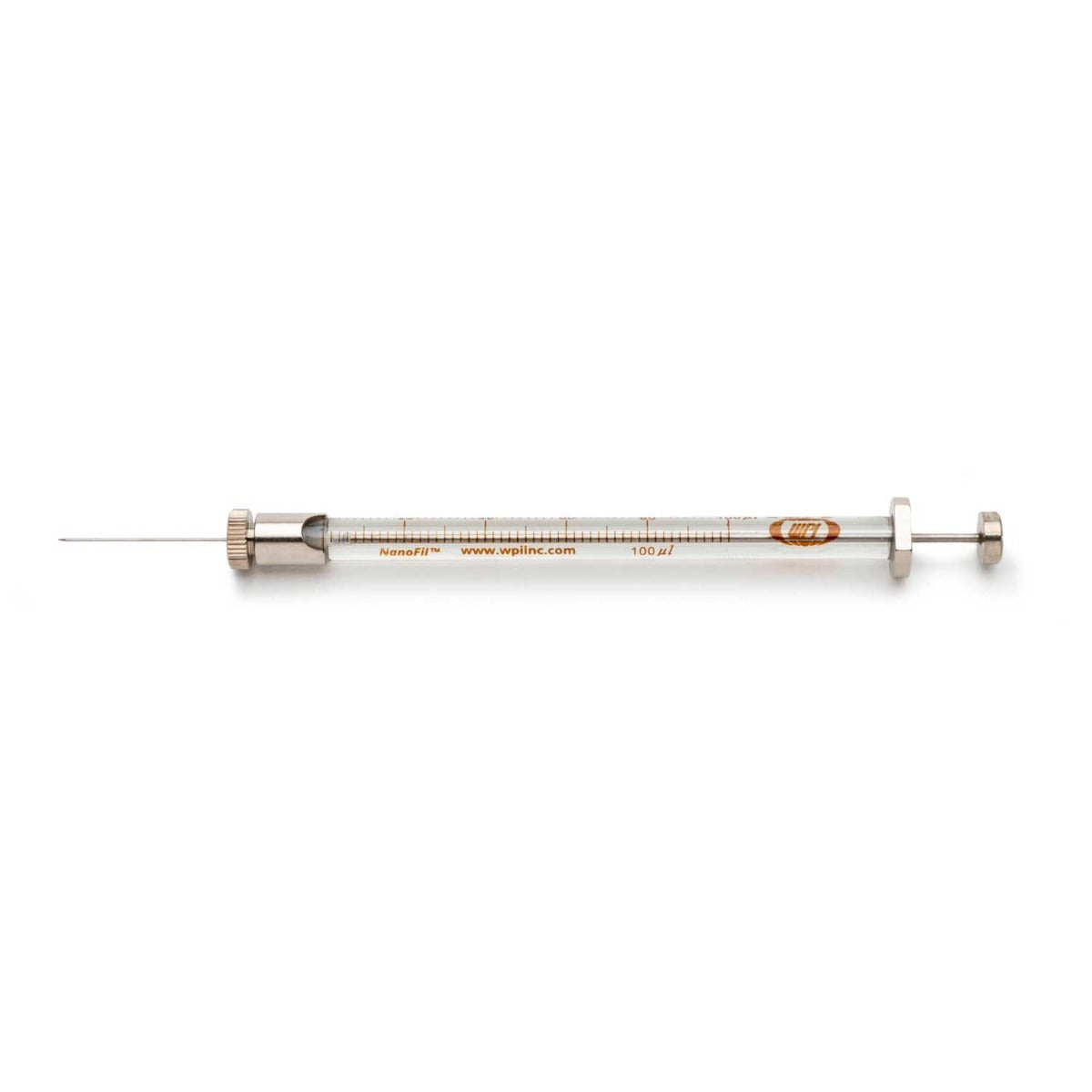
The NanoFil™ Gas-Tight Injection System boasts zero dead volume, an the variability is eliminated with the NanoFil™ design: our interchangable needles advance directly into the syringe barrel, creating a 1:1 connection with the plunger— a truly gas-tight system built for low-volume sample control. Our cross-compatible NanoFil™ needles are offered in sizes from 33-36g, blunt or beveled.
* Returns Policy : Non-returnable/Non-refundable
Visit the Gas-Tight Injection System page to learn about its gas-tight design, needle versatility, etc.
| Order code | Size | Includes |
| NANOFIL | 10 μL |
|
| NANOFIL-100 | 100 µL |
|

Try our quartz 34G flexible tubing for precision capillary backfilling (SKU #NFQ34-5). Particularly useful for low-volume sample transfer into long taper glass pipettes. Compatible with any NanoFil™ syringe!





Retinal Pigment Epithelium (RPE) Injection Kit & Intra Occular (IO) Injection Kit
How to select the correct tip for your application.
See why our system is the choice for low-volume applications, and why other designs are not dependable: Tired of Missing the Mark?
Note: This product is non-returnable/non-refundable. For details, please refer to the WPI Terms & Conditions.
*After repeated use, it is normal that your plunger may incur wear/tear to either the PTFE tip and/or its overall straightness. The syringe must be replaced as plungers are specially made to each syringe.
NanoFil™ Gas-Tight Injection System Manual
NanoFil™ Gas-Tight Injection System Spec Sheet
Selecting Your NanoFil™ Needle Tip
How to Prime Your NanoFil Gas-Tight Syringe System
Unboxing Your NanoFil & Installing a Needle
In the video below, you can see how to front fill an Nanofil syringe.
How to Choose the Correct NanoFil Tip Size for Microinjections
How to Use the NanoFil Syringe for Retinal Pigment Epithelial and Intraocular Injections
How to Install a NanoFil Syringe on an UltraMicroPump
How to Setup the NanoFil System for Making Injections
NanoFil Microliter Syringes for Accurate Microinjections
NanoFil Needle Cause Less Tissue Trauma By Design
Guo, Q., Gobbo, D., Zhao, N., Zhang, H., Awuku, N.O., Liu, Q., Fang, L.P., Gampfer, T.M., Meyer, M.R., Zhao, R., Bai, X., Bian, S., Scheller, A., Kirchhoff, F., & Huang, W. (2024). Adenosine triggers early astrocyte reactivity that provokes microglial responses and drives the pathogenesis of sepsis-associated encephalopathy in mice. Nature Communications, 15; 6340. https://doi.org/10.1038/s41467-024-50466-y
Luo, S., Jiang, H., Li, Q., Qin, Y., Yang, S., Li, J., Xu, L., Gou, Y., Zhang, Y., Liu, F., Ke, X., Zheng, Q., & Sun, X. (2024). An adeno-associated virus variant enabling efficient ocular-directed gene delivery across species. Nature communications, 15(1), 3780. https://doi.org/10.1038/s41467-024-48221-4
Nam, J., Richie, C.T., Harvey, B.K., & Voutilainen, M.H. (2024). Delivery of CDNF byAAV‑mediated gene transfer protects dopamine neurons and regulates ER stress and inflammation in an acute MPTP mouse model of Parkinson’s disease. Communications Biology, 7; 966. https://doi.org/10.1038/s42003-024-06658-9
Wietek, J., Nozownik, A., Pulin, M., Saraf-Sinik, I., Matosevich, N., Gowrishankar, R., Gat, A., Malan, D., Brown, B.J., Dine, J., Imambocus, B.N., Levy, R., Sauter, K., Litvin, A., Regev, N., Subramaniam, S., Abrera, K., Summarli, D., Goren, E.M., Mizrachi, G., Bitton, E., Benjamin, A., Copits, B.A., Sasse, P., Rost, B.R., Schmitz, D., Bruchas, M.R., Soba, P., Oren-Suissa, M., Nir, Y., Wiegert, J.S., & Yizhar, O. (2024). A bistable inhibitory optoGPCR for multiplexed optogenetic control of neural circuits. Nature Methods, 21; pg. 1275–1287. https://doi.org/10.1038/s41592-024-02285-8
Zhang, C., Dulinskas, R., Ineichen, C., Greter, A., Sigrist, H., Li, Y., Alanis-Lobato, G., Hengerer, B., & Pryce, C.R. (2024). Chronic stress deficits in reward behaviour co-occur with low nucleus accumbens dopamine activity during reward anticipation specifically. Communications Biology, 7; 966. https://doi.org/10.1038/s42003-024-06658-9

| Tip Order Number | Tip O.D. (µm) | Tip I.D. (µm) | Tip Length (mm) | Total Length (mm) | Shank O.D. (µm) | Bevel Length (µm) | Tip Material |
| NF33BV | 210 | 115 | 10 | 40 | 460 | ≈348 | Stainless Steel |
| NF34BV | 185 | 85 | 5 | 35 | 460 | ≈290 | Stainless Steel |
| NF35BV | 135 | 55 | 5 | 35 | 460 | ≈204 | Stainless Steel |
| NF36BV |
120 |
35 | 3 | 33 | 460 | ≈156 | Stainless Steel |
| NFQ34-5 | 160 | 100 | 55 | 75 | 460 | Quartz | |
| NF33BL | 210 | 115 | 10 | 40 | 460 | ≈0 | Stainless Steel |
| NF34BL | 185 | 85 | 5 | 35 | 460 | ≈0 | Stainless Steel |
| NF35BL | 135 | 55 | 5 | 35 | 460 | ≈0 | Stainless Steel |
| NF36BL | 120 | 35 | 3 | 33 | 460 | ≈0 | Stainless Steel |
| Silflex | 100 | 35 | |||||
| NF26BV | 460 | 140 | 40 | 460 | Stainless Steel |
NANOFIL™ Syringe Specification Table
Syringe Type: Gas-tight
Syringe Body: Borosilicate glass, Stainless Steel, PTFE
Autoclavable/Gas Sterilizable:
Package Quantity: 1 syringe
| Syringe Order Code | Volume | Plunger Stroke Length |
Plunger Cap Dimensions |
Syringe Barrel I.D. / O.D.* |
Syringe Type Selection |
| NANOFIL |
10µL |
60mm |
O.D.*: 7.90mm (0.311in) Depth: 2.80mm (0.110in) |
0.46mm (0.018in)/ 6.40mm (0.252in) |
L (MICRO4) 5 (MICRO2T) |
| NANOFIL-100 | 100µL | 0.46mm (0.018in)/ 6.40mm (0.252in) |
*I.D. = Inner diameter, O.D. = Outer diameter





$140.00

NF26BV-2


Multiple SKUs

NFGSK-5



NFQ34-5


Multiple SKUs






In this video, Mike Pizza demonstrates how to front fill a Nanofil syringe using MicroFil.
WPI’s NanoFil™ is a gas-tight microliter injection system for small-animal research that supports needles down to 36 gauge. Its ultra-low dead volume enables direct sub-microliter injections without oil backfilling, and a patented silicone gasket allows quick needle switching with minimal sample loss. Compatible with GC/CE capillaries and various tubing, it offers blunt and unique 25° tri-surface beveled needles (26–36G) that reduce tissue damage and improve durability. The system is widely used for precise tissue injections, including ophthalmic applications, and is supported by application kits and peer-reviewed studies.
Application kits are specially designed for eye research for injecting retinal pigment epithelium (RPE) and for intraocular (IO) injections. In addition, these kits may be used for brain injection in mice. They need to be used with a NanoFil syringe and UMPIII to achieve accurate, repetitive and oil-free injection in the submicroliter range.
The system depicted includes components often favored by researchers:
Syringes have a wide range of applications in a laboratory setting due to their ability to accurately measure and dispense liquids. Common lab uses include transferring precise volumes of liquid samples from one container to another for dilution, mixing, reagent preparation, and sample extraction. You may be using them for analytical techniques, like chromatography (such as high-performance liquid chromatography (HPLC) or gas chromatography), spectrophotometry, and titration. Syringes are often used for precise dosing and delivery of reagents during chemical reactions and synthesis processes, or in life science research for aspirating media, adding growth factors or nutrients, and transferring cells. In medical and veterinary research, syringes are used for the injection or infusion of drugs, vaccines, or contrast agents into animal models or cell cultures. Researchers can use lab syringes to collect liquid samples, particularly in environmental monitoring, water quality analysis, or biological sample collection. In microfluidic applications and lab-on-a-chip systems, syringes are perfect for precision control of fluid volumes and flow rates.
Selling fast!
Get yours while you can.
