This website uses cookies to ensure you get the best experience on our website.
Read more
Biosensors
Selection includes high selectivity and low detection limit sensors
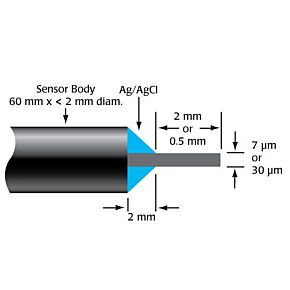
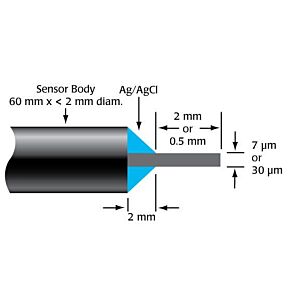
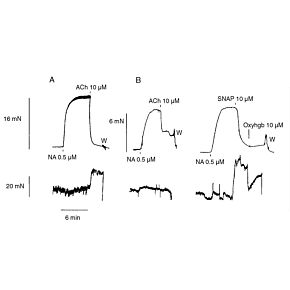
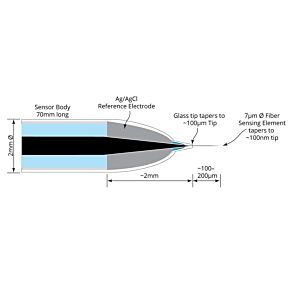
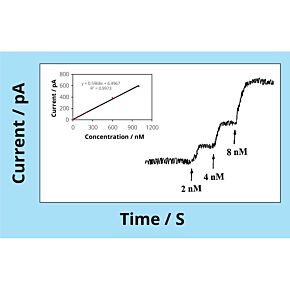
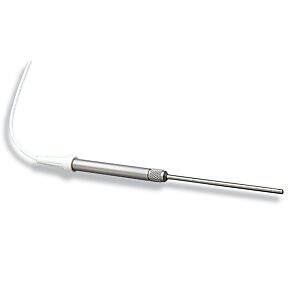
WPI's biosensors are unique, because they offer a high selectivity and low detection limit (down to nM concentration) with a broad dynamic range, covering physiological concentrations of species with different sizes from nanometer to millimeter. The majority of our sensors are the only commercially available sensors in the world. Scientists across a variety of disciplines have relied on our sensors for over 25 years. These scientists use WPI's sensors for research performed in universities, hospitals, biomedical research labs, pharmaceutical companies, food/nutrition research labs, environmental monitoring centers and military labs. Our popular biosensors are listed in thousands of publications.
- hiddenBack to TopClose