Selling fast!
Get yours while you can.
Free Shipping on web orders over $50! (USA only. Some exclusions.) Free Shipping on web orders over $50! (USA only. Some exclusions.)























KWIK-SIL
Couldn't load pickup availability
Prices valid in USA, Canada, and PR only.
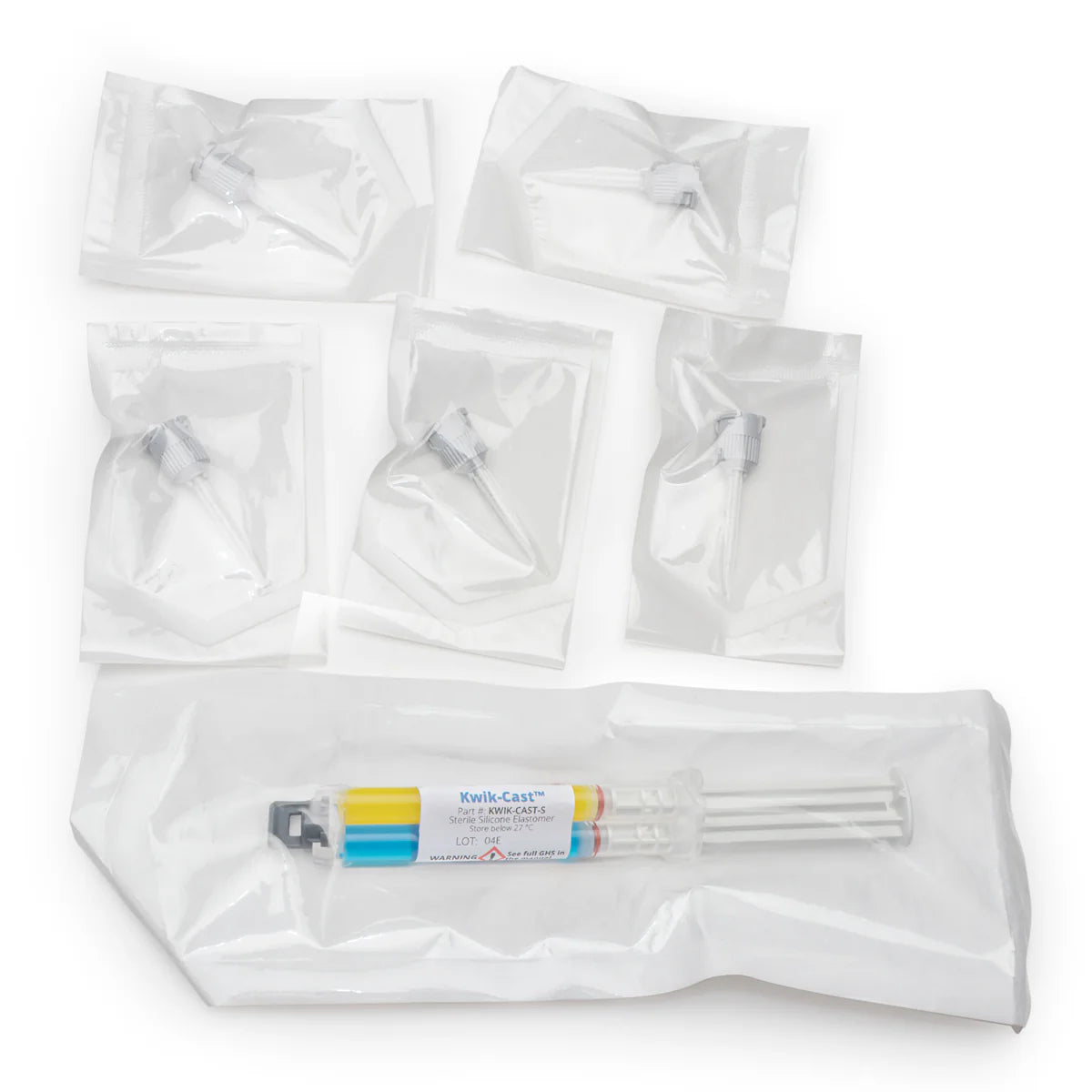
Kwik-Sil™ (adhesive) and Kwik-Cast™ (sealant) are low-toxicity, biocompatible silicone elastomers designed for neuroscience, nerve studies, and live tissue applications. Delivered in a dual-barrel cartridge, the materials mix automatically during application, eliminating pre-mixing and simplifying workflows.
They cure rapidly at room temperature without generating heat, reducing the risk of tissue damage while maintaining consistent, reproducible results. Kwik-Sil™ dries clear and cures slightly faster for adhesion, while Kwik-Cast™ dries green and flows easily for sealing and embedding. Sterile options are available.
* New and Improved with Extended Shelf Life
* Returns Policy: Non-returnable/Non-refundable
COMPARE OPTIONS FOR YOUR PROTOCOL

| Feature | Kwik-Sil™ (Adhesive) | Kwik-Cast™ (Sealant) |
|---|---|---|
| Primary use | Adhesion | Sealing / embedding |
| Viscosity | Medium | Very low (flows easily) |
| Color | Clear | Green (when color mixes) |
| Cure speed | Faster | Slightly slower (~3 min) |
| Standard Pkg | 2 Dual Barrel Cartridges with 10 Mixing Tips |
2 Dual Barrel Cartridges with 10 Mixing Tips |
| Sterile Pkg | 1 Sterile Dual Barrel Cartridge with 5 Mixing Tips | 1 Sterile Dual Barrel Cartridge with 5 Mixing Tips |
| Best for | Chronic studies, electrode fixation | Acute recordings, nerve embedding |

Kwik-Sil™ and Kwik-Cast™ are formulated for use with live tissue, offering significantly lower toxicity than traditional RTV silicones. During curing, they release a small amount of hydrogen gas rather than acetic acid or alcohol, reducing the risk of cellular damage.
Both materials cure within minutes at room temperature without generating heat and are not inhibited by contact with biological tissue. This allows for reliable performance in applications such as electrode fixation, nerve embedding, and in vivo experimentation.
The result is a fast, reproducible silicone system that supports consistent outcomes across sensitive biological workflows.
Kwik-Sil™ is best suited for applications that require stable adhesion over time, such as chronic peripheral nerve studies and long-term electrode fixation.
Kwik-Cast™ is designed for embedding and sealing in acute recordings. It flows around tissue and electrodes to create a continuous seal that prevents fluid intrusion and supports stable signal acquisition. It can also be applied under mineral oil and allows easy electrode recovery after use.
Choose Kwik-Sil™ for adhesion and durability in longer experiments, and Kwik-Cast™ for sealing, embedding, and short-term recording setups.
Kwik-Sil™ and Kwik-Cast™ cure within minutes at room temperature, providing a short, reliable working time for precise application.
The silicone inside the cartridge cannot be sterilized by UV, gamma, H₂O₂, autoclaving, or EtO. For sterile workflows, use the pre-sterilized versions (KWIK-SIL-S™ and KWIK-CAST-S™).
The exterior of the cartridge and mixing tips can be cleaned using 70/30 alcohol or liquid sterilants such as Cidex OPA (WPI P/N 7364-4) or Rapicide OPA (WPI P/N 504611).

Kwik-Sil™ and Kwik-Cast™ come with a tip design engineered to completely mix the adhesive and minimize leakage.
NOTE: This product is non-returnable/non-refundable. For details, please refer to the WPI Terms & Conditions.
KWIK-SIL/KWIK-CAST Instruction Manual
5 Reasons KWIK-SIL & KWIK-Cast Adhesive are Ideal for Neuroscience Application
KWIK-SIL versus KWIK-Cast, What's the Difference
KWIK-SIL & KWIK-CAST: How to Install the Mixing Tip
| Type | KWIK-SIL | KWIK-SIL-S | KWIK-CAST | KWIK-CAST-S |
| MIX RATIO | 1 to 1 | 1 to 1 | 1 to 1 | 1 to 1 |
| WORKING TIME | < 5 minutes* | < 5 minutes* | 4 minutes | 4 minutes |
| SETTING TIME (ROOM TEMP., 1:1 RATIO) | 5–10 minutes** | 5–10 minutes** | <10 minutes | <10 minutes |
| CURE TIME | ~15 minutes | ~15 minutes | ~15 minutes | ~15 minutes |
| VISCOSITY, CPS | 15,000 | 15,000 | 10,000 | 10,000 |
| GUARANTEED SHELF LIFE AT 23 °C | 6 months | 6 months | 6 months | 6 months |
| VOLUME | 5 mL | 5 mL | 5 mL | 5 mL |
| NUMBER OF MIXING TIPS | 10 | 5 | 10 | 5 |
| DEAD VOLUME OF THE MIXING TIP | <0.12 mL | <0.12 mL | <0.12 mL | <0.12 mL |
| NOTE | This is the consistency of pourable silicone rubber. | This is the consistency of pourable silicone rubber. | This is the consistency of chocolate syrup. | This is the consistency of chocolate syrup. |
| AFTER CURING 24 HOURS: | ||||
| TEAR STRENGTH, PPI | 90 | 90 | 44 | 44 |
| ELONGATION % | 650 | 650 | 60 | 60 |
| DUROMETER (SHORE A-2) | 30 | 30 | 36 | 36 |
| COLOR | translucent | translucent | green | green |
| VOLUME RESISTIVITY, W/CM | 1x1015 | 1x1015 | 1x1015 | 1x1015 |
* 3 minutes average with about 90 seconds of liquidity
** no longer mixable at this point
Bubbles are caused by hydrogen gas released during curing. Because the material cures quickly, the gas may not fully escape. To reduce bubbles, cool the material before use to slightly slow the curing process.
No. The silicone inside the cartridge cannot be sterilized by UV, gamma radiation, H₂O₂, autoclaving, or EtO. For sterile applications, use the pre-sterilized versions (KWIK-SIL-S or KWIK-CAST-S).
KWIK-SIL is designed for adhesion and long-term stability, making it ideal for chronic studies and electrode fixation. KWIK-CAST is formulated for sealing and embedding, flowing easily around tissue for acute recordings and short-term experiments.
Working time is typically under 5 minutes, with curing completed in approximately 10–15 minutes at room temperature.
Yes. Once cured, the material can be gently removed. For example, it can be peeled away from rodent fur or tissue with minimal disruption when handled carefully.






















$137.00

1571


503763


VETBOND


600022
When you are looking for a low toxicity adhesive with some elasticity and good moisture resistance, choose a silicone adhesive.
Both Kwik-Sil and Kwik-Cast are silicone adhesives, but they are a little different.
The adhesive you select will depend largely on your biomedical research laboratory application. If you are building electrodes, you might use a silver-filled, conductive epoxy to connect a Ag/AgCl pellet. For instantaneous bonding when mounting a rodent brain slice, you might select an ethyl cyanoacrylate. When you are looking for a low toxicity adhesive with some elasticity and good moisture resistance, a silicone adhesive is the option of choice. When used with living tissue, an adhesive must be:
In any laboratory, having key lab supplies is almost as important as having the major equipment. Choosing a reputable supplier of these necessary supplies is as important as having quality laboratory supplies when you need them. WPI wants to be your partner in early drug discovery, and we stock a wide variety of lab supplies, many of which can ship the same business day. Having a variety of lab supplies ready to ship makes us a dependable research partner. Here are some of the popular supplies that we keep on hand to meet your needs for your upcoming experiment
If you’ve ever stepped into a research lab, you know it’s filled with specialized tools and equipment—all essential for keeping experiments running smoothly. When people ask “What are laboratory supplies” the short answer is: they’re the essentials that help scientists and researchers get their work done. That includes everything from basic items like petri dishes and pipettes to more advanced laboratory equipment like microscopes and micromanipulators.
Selling fast!
Get yours while you can.
