Selling fast!
Get yours while you can.
Free Shipping on web orders over $50! (USA only. Some exclusions.) Free Shipping on web orders over $50! (USA only. Some exclusions.)





MF28G-5
Couldn't load pickup availability
Prices valid in USA, Canada, and PR only.
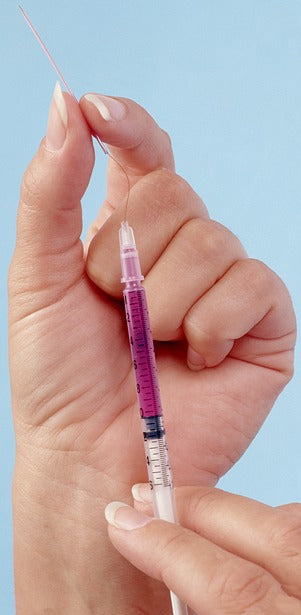
Our MicroFil fills micropipettes easily and reliably. Its long and fine tip allows you to start the filling very close to the pipette tip, eliminating both air bubble formation and clogging due to the washing down of dust particles. The transparent amber MicroFil needle is constructed from a combination of plastic and fused silica - no metal components are used. The MicroFil plastic needle may be stored for days with the filling solution inside without clogging. Sold in packages of 5.
Custom MicroFil cut to your desired length is also available.
| Order code | Gauge | ID (µm) | OD (µm) | Length (mm) |
| MF34G-5 | 34 | 100 | 164 | 67 |
| MF28G-5 | 28 | 250 | 350 | 97 |
| MF28G67-5 | 28 | 250 | 350 | 67 |
Our MicroFil fills micropipettes easily and reliably. Its long and fine tip allows you to start the filling very close to the pipette tip, eliminating both air bubble formation and clogging due to the washing down of dust particles. The transparent amber MicroFil needle is constructed from a combination of plastic and fused silica - no metal components are used. The MicroFil plastic needle may be stored for days with the filling solution inside without clogging. Sold in packages of 5.
The MicroFil's tip elasticity is sturdy and very flexible though not unbreakable. Since it is more flexible than stainless steel needles, moderate bending will not block or damage the MicroFil needle. The combination of plastic and fused silica in the MicroFil tip is sturdier than plastic tips, allowing easy and repeated insertions into micropipettes. MicroFil's luer fitting allows easy coupling to syringes and syringe filters.
MicroFil is constructed of fused silica, coated with Polyimide. The luer fitting is HDPP and is held in place with a medical grade UV adhesive.
NOTE: The fused silica tubing is susceptible to water intrusion that makes it more fragile. Pressurized steam increases this fragility.
Chowdhury, S. A., & Rasmusson, D. D. (2002). Comparison of receptive field expansion produced by GABAB and GABAA receptor antagonists in raccoon primary somatosensory cortex. Experimental Brain Research, 144(1), 114–121. http://doi.org/10.1007/s00221-002-1035-7
Bito, V., Sipido, K. R., & Macquaide, N. (2015). Basic methods for monitoring intracellular Ca2+ in cardiac myocytes using Fluo-3. Cold Spring Harbor Protocols, 2015(4), 392–7. http://doi.org/10.1101/pdb.prot076950




$84.00


Multiple SKUs
In any laboratory, having key lab supplies is almost as important as having the major equipment. Choosing a reputable supplier of these necessary supplies is as important as having quality laboratory supplies when you need them. WPI wants to be your partner in early drug discovery, and we stock a wide variety of lab supplies, many of which can ship the same business day. Having a variety of lab supplies ready to ship makes us a dependable research partner. Here are some of the popular supplies that we keep on hand to meet your needs for your upcoming experiment
Selling fast!
Get yours while you can.
