This website uses cookies to ensure you get the best experience on our website.
Read more
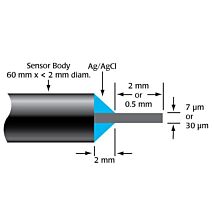
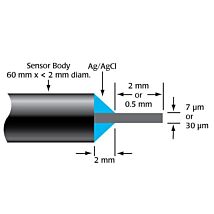
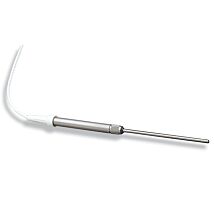
2mm SHIELDED HPO SENSOR
$1,550.00
Prices valid in USA, Canada, and PR only.
Order code
ISO-HPO-2

Prices valid in USA, Canada, and PR only.
Direct quantitative measurements of hydrogen peroxide in biological samples
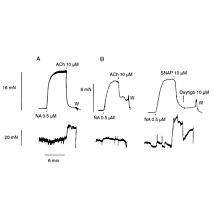
Despite the recognized importance of this oxidant in biology, real-time measurements at low concentration have been difficult. The hydrogen peroxide sensors developed by WPI are designed to compliment existing high sensitivity fluorescent approaches with direct quantitative measurement in biological samples in the low nM range.
Prices valid in USA, Canada, and PR only.
See the latest Biosensor Data Sheet.
Features
- Gas permeable polymer membrane sleeve blocks liquids, ions and particulate matter
- Incorporated reference electrode
- For use with Apollo1000, Apollo4000, TBR4100 and TBR1025
- Package of 1
Benefits
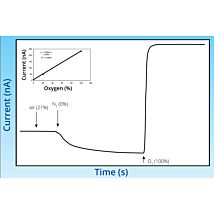
- Response Time: < 5 s (90%)
- Detection Limit: < 100 nM to 100 μM (100 mM custom)
- Drift: < 0.2 pA/min
- Sensitivity: 8 pA/μM
Applications
- Cell Cultures
The ISO-HPO-2 is a 2.0 mm stainless steel sensor, with replaceable membrane sleeves (600012) and an internal refillable electrolyte (100042). It is designed for use in cell cultures and similar applications.
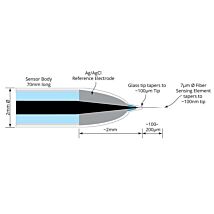
The ISO-HPO-100 is a 100 µm tip diameter hydrogen peroxide micro sensor designed for use in tissues and similar applications. The design is based on a platinum wire sensing electrode coated with a proprietary membrane to enhanceH2O2 detection.
These sensors incorporate WPI’s proprietary combination electrode technology whereby the hydrogen peroxide sensing element and separate reference electrode are encased within a single Faraday-shielded probe design. This design has been shown to enhance performance during measurements and minimizes overall sensor size.
Our hydrogen peroxide (H2O2) sensors work with the TBR4100 and TBR1025 free radical analyzers.
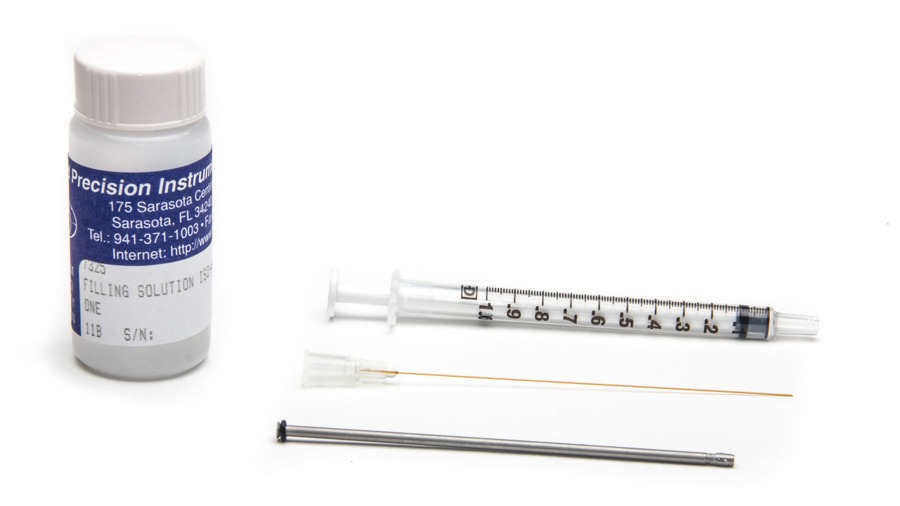
NOTE: The 2mm biosensors come with a spare membrane sleeve, 1 cc syringe, an MF28G67 MicroFil needle and 10 mL of filling solution.
Hydrogen peroxide in biological systems
Hydrogen Peroxide is produced in biological systems by controlled pathways at low concentrations that impact on cell signaling. At higher concentrations inflammatory cells produce local intense amounts of this oxidant to kill pathogens. In the progress of human disease, uncontrolled formation of hydrogen peroxide from the mitochondrial respiratory chain and enzymes, such as xanthine oxidase, can occur (Prof. Victor Darley-Usmar, Univ. of Alabama, personal communication).
| SKU | ISO-HPO-2 |
|---|
Upsell Products
Olson, K. R., Gao, Y., DeLeon, E. R., Arif, M., Arif, F., Arora, N., & Straub, K. D. (2017). Catalase as a sulfide-sulfur oxido-reductase: An ancient (and modern?) regulator of reactive sulfur species (RSS). Redox Biology, 12, 325–339. https://doi.org/10.1016/j.redox.2017.02.021
DeLeon, E. R., Gao, Y., Huang, E., Arif, M., Arora, N., Divietro, A., … Olson, K. R. (2016). A case of mistaken identity: are reactive oxygen species actually reactive sulfide species? American Journal of Physiology-Regulatory, Integrative and Comparative Physiology, 310(7), R549–R560. https://doi.org/10.1152/ajpregu.00455.2015
Orellano, L. A. A., Almeida, S. A., Campos, P. P., & Andrade, S. P. (2015). Angiopreventive versus angiopromoting effects of allopurinol in the murine sponge model. Microvascular Research, 101, 118–126. https://doi.org/10.1016/j.mvr.2015.07.003
Pandolfi, C., Pottosin, I., Cuin, T., Mancuso, S., & Shabala, S. (2010). Specificity of Polyamine Effects on NaCl-induced Ion Flux Kinetics and Salt Stress Amelioration in Plants. Plant and Cell Physiology, 51(3), 422–434. https://doi.org/10.1093/pcp/pcq007