This website uses cookies to ensure you get the best experience on our website.
Read more
Free Radical Analyzers
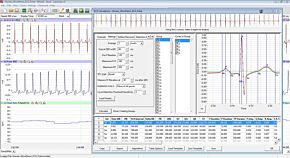
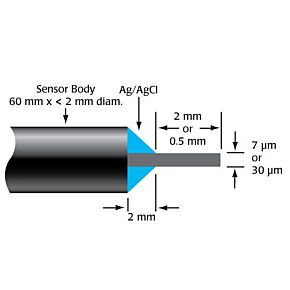
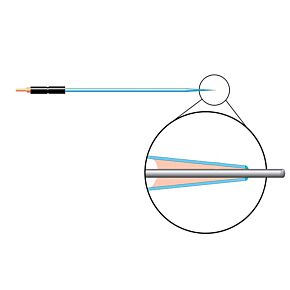
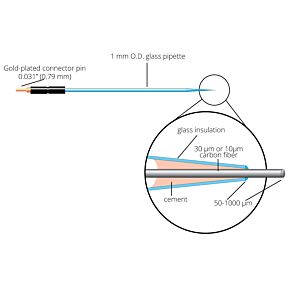
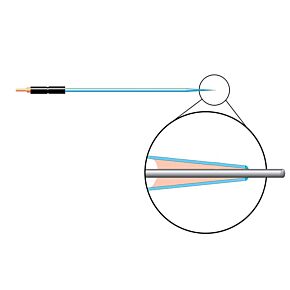
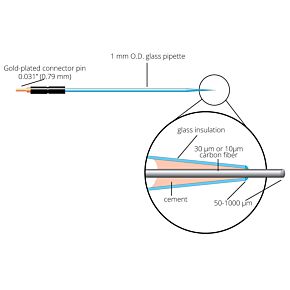
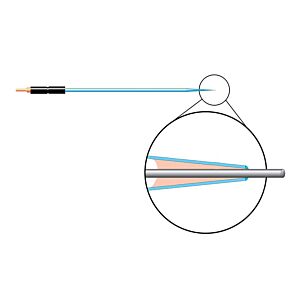
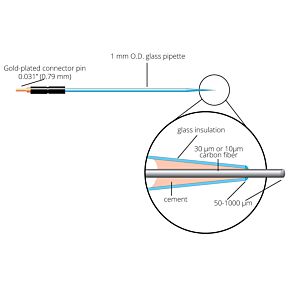
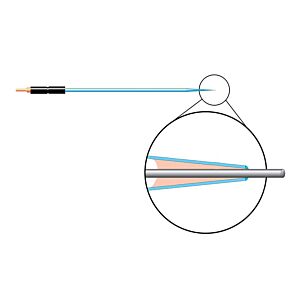
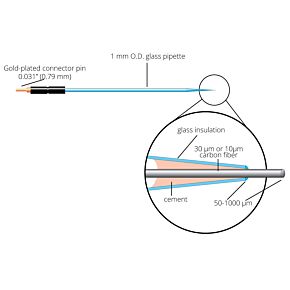
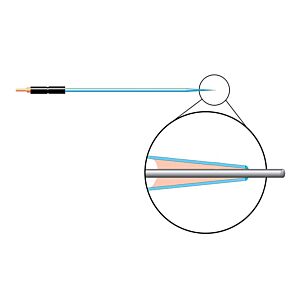
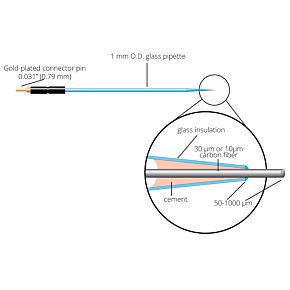
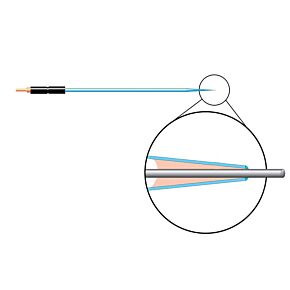
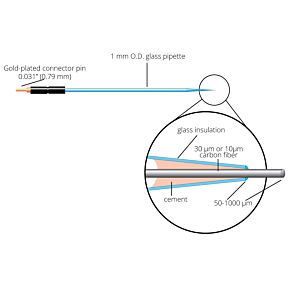
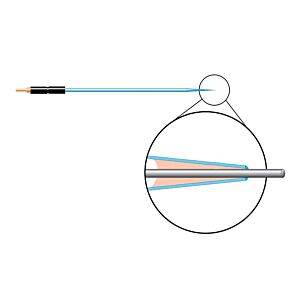
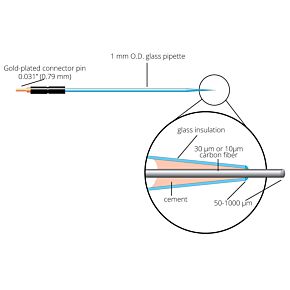
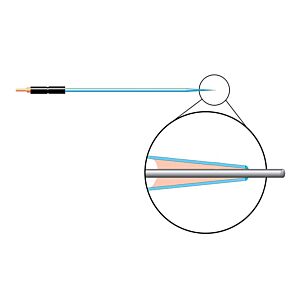
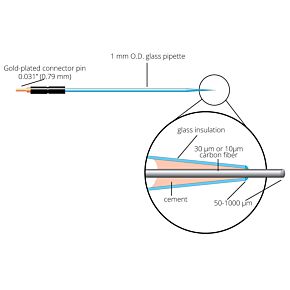
Our free radical analyzers are used in biosensing experiments to detect oxygen, nitric oxide, carbon dioxide, hydrogen peroxide, hydrogen sulfide and glucose, using WPI ion-species specific biosensors. Our TBR series biosensing analyzers can be set to monitor the specific species of interest, and the 4-channel TBR4100 can monitor four biosensors simultaneously. While the TBR biosensing device is commonly used as a nitric oxide analyzer, it is perfect for real-time detection and measurement of a variety of redox-reactive species using the electrochemical (amperometric) detection principle. This series of optically isolated biosensing analyzers has ultra low noise and independently operated channels
- hiddenBack to TopClose