This website uses cookies to ensure you get the best experience on our website.
Read more
EVOM or Volt Meter: Defining the Difference
April 24, 2013

 A Volt Meter
A Volt Meter
A volt-ohm meter may apply a constant voltage of unknown current across the membrane and damage the cells electrically and also leave a chemical imbalance in the electrodes. WPI experimented with two volt-ohm meters:
- An expensive Fluke meter applies a DC voltage of 50 to 80mV, which (in a 1000Ω membrane) will pass 80µA. With a 200Ω membrane, this is 400µA.
- A discount meter puts out 500mV DC, which will pass 500µA through the membrane. With a 200Ω membrane, this is 2.5mA.
Volt Meter Charges or Electrocutes Cells
When you use a volt ohm meter to test a cell layer, the Ag/AgCl electrodes accumulate an unbalanced chemical charge. If the cells are not electrocuted in the process, they also accumulate a charge. The unbalanced electrodes can now act as a source of voltage and current that has to be overcome to make an accurate measurement.
If a volt ohm meter was employed in a TEER (Trans Epithelial Electric Resistance) measurement and left measuring the cells for more than a few seconds, then the TEER measurement value would probably drift downwards as the cells and electrodes are changed by the voltage and current applied. In a perfect world, a measuring device minimizes its impact on what it is attempting to measure. These meters are best left to measuring fixed resistors and circuits.
EVOM - Perfect for TEER
Reversing Polarity
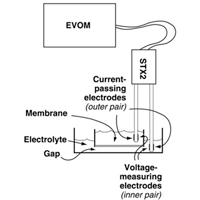
The EVOM² passes a constant current of 10µA through membrane and reverses the polarity 12.5 times per second so that it does not leave a charge behind on either the electrodes or the membrane. The voltage that is present at 10µA on 1000Ω is 10mV. (On a side note, even this is too much for some tissues, like retinal.) Typically, this value on a 200Ωmembrane is 50µV. At either setting, that is much less energy to dissipate in the membrane or to charge the electrodes.
The EVOM² passes the 12.5Hz constant current 10µA signal through the two current electrodes (I1 and I2) on the STX through the membrane. The companion electrodes (V1 and V2) measure the voltage that was required to reach the 10µA current and sends this to the processor. The processor converts this to ohms via Ohm's Law (E=I/R) and displays the signal on the digital meter. Since the current is fixed at 10µA, the processor can easily convert the measurement.
Built in Averaging
The EVOM (a legacy product replaced by the EVOM2) and EVOM² both have averaging built in so that spurious readings are “sampled out.” The EVOM² has a recording output connector so that it can be fed into a data recorder. The EVOM² system requires the use of a conductive liquid to make the measurement. If the STX electrodes are in non-conductive air, the reading is invalid. The older model had an alarm in it to indicate a broken electrode or measurements in air or non-conductive media.
Electrode Stabilizing for Balancing
The EVOM² has an electrode stabilizing function built into the meter. When the meter is powered off, the V1 and V2 electrodes are short-circuited together so that if the researcher places them in a conductive liquid, then those electrodes are balanced at 0mV. That is, they are “equilibrated.” This electrode balancing is critical to membrane potential measurements. It is not as critical with TEER measurements, because the EVOM² electronics are specifically designed to compensate for electrode imbalances. The EVOM² is recognized worldwide as a standard measuring devices for this science.
Summary
The EVOM² qualitatively measures cell monolayer health and quantitatively measures cell confluence. The EVOM² is best suited for TEER measurement and monolayer confluence detection.

Close